Electrochemical machining is developed by W. Gussef in 1929. In this process, the material removal takes place using an Electrochemical process.
This machining process is usually used to remove metal from extremely hard materials or materials that are difficult to machine using conventional processes.
Want to know about the electrochemical machining process, from scratch to end? Then, you landed in the right place.
Rest assured, you will get a complete guide on the ECM machining process from basic to advanced. So, sit back and relax while I start the engine of Engineers Rail.
Before I start, Here is the bonus for you. At the end of the article, You can download this whole article as a PDF and PPT, and enjoy it offline, whenever you need it.
So, without further delay, let’s dive in-
What is Electrochemical Machining?-
It is the method of removing metal from the electrochemical process, where DC current is passed between the tool(cathode) and workpiece(Anode) through an electrolyte, known as ECM machining.
In this process, a combination of electrical energy and chemical energy is used to remove materials from the workpiece.
The ECM technique is most frequently used to create complex geometries, such as turbine blades, with an excellent surface polish from materials that are challenging to machine.
Additionally, it is commonly and successfully used as a deburring method.
Working Principle of Electrochemical Machining-
Electrochemical machining works based on Faraday’s laws of electrolysis.
The amount of metal removed by machining or deposited is calculated from Faraday’s laws of electrolysis, Which states that-
The amount of mass removed by machining, m, is directly proportional to the amount of electricity.
m ∝ It
The amount of different substances dissolved, m, by the same quantity of electricity(It) is proportional to the substance’s chemical equivalent weight.
m ∝ ∈
Where ∈ = Chemical equivalent weight.
Here,
ECM uses direct current at a high density of 0.5 to 5 A/mm² and a low voltage of 10 to 30V.
The machining current passes through the electrolytic solution that fills the gap between an anodic workpiece and a pre-shaped cathodic tool.
The electrolyte is forced to flow through the interelectrode gap at a high velocity, usually more than 5m/s to intensify the mass and charge transfer through the sublayer near the anode.
The electrolyte removes the dissolution products, such as metal hydroxide, heat, and gas bubbles, generated in the interelectrode gap.
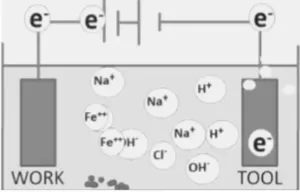
The example of the dissolution reaction of iron in sodium chloride(NaCl) water solution as an electrolyte.
The result of electrolyte dissolution and Nacl dissolution leads to-

The negative charge anions OH– and Cl– moved towards the anode and positively charged cations of H+ and Na+ are directed to the cathode. At the anode, Fe changes to Fe++ by losing two electrons.
Fe→ Fe++ + 2e
At the cathode, the reaction involves the generation of hydrogen gas and hydroxyl ions.
2H2O+2e→H2 +2(OH)–
The outcome of these electrochemical reactions is that iron ions combine with other ones to precipitate out as iron hydroxide, Fe(OH)2
Fe+2H2O→Fe(OH)2+H2
The ferrous hydroxide may react further with water and oxygen to form the ferric hydroxide, Fe(OH)3
4Fe(OH)2+2H2O+O2→4Fe(OH)3
With this metal electrolyte combination, electrolysis has involved the dissolution of iron, from the anode and the generation of hydrogen, at the cathode.
Electrochemical Machining Parts & Construction-
There are several electrochemical machining equipment used to construct the electrochemical machine in the electrochemical machining process–
- Dc power supply
- Table
- Workpiece
- Tool
- Feed Unit
- Electrolyte
- Pump
- Filter
- Pressure gauge
- Flow Meter
1. DC power supply-
The DC power supply used in this process is from 3 to 30V.
The DC power supply is used to create a potential difference between the workpiece and the tool and make them act as anode and cathode respectively.
2. Table-
The table used to hold the workpiece on the top of it.
3. Workpiece-
This is the object from which material is to be removed. The workpiece is connected to the positive terminal of the DC power supply and acts as an anode.
4. Tool-
This tool is connected to the negative terminal and acts as a cathode. The too(cathode) should have good thermal conductivity and electrical conductivity and also should be corrosion-proof.
The most commonly used tools in the ECM machining process are stainless steel, brass, copper, etc.
5. Feed Unit-
It is used to constantly move the tool downward towards the workpiece.
6. Electrolyte-
The electrolyte is used to establish an electrical connection between the anode and the cathode so that positive and negative ions flow from these two.
The electrolyte is used to remove the heat produced from the passage of the current.
The most commonly used electrolyte used in the ECM process is salt electrolytes like sodium chloride and sodium nitrate.
7. Pump-
This pump is used to pump the electrolyte towards the gap between the cathode and anode.
8. Filter-
The filter is used to filter the electrolyte and remove the unwanted particles from the electrolyte.
9. Pressure gauge-
The pressure gauge is used to control the pressure at which the electrolyte is supplied to the system of electrochemical machining.
10. Flow Meter-
Flowmeter is used to regulate the flow of electrolyte which is supplied to the system.
⇒Here, After knowing various equipment used in ECM, Now, I am heading to working of the same.
So, stay with me and scroll gently..,
Working of Electrochemical Machining-
- The electrolyte is supplied to the system of anode and cathode, ie, workpiece and tool using the pump.
- In between the pump and the system a filter is placed which is used to filter the electrolyte before reaching the system.
- After passing through the filter and flow regulator, the electrolyte reaches the space between the workpiece and the tool.
- As the electrolyte reaches the gap between anode and cathode, electrical contact is established between the anode and the cathode.
- After the electrical contact is established, the positive ions start flowing from the workpiece towards the tool and negative ions start flowing from the tool towards the workpiece.
- When the positive ions flow from the workpiece towards the tool, the electrolyte carries away the positive ions with it and stops it from reaching the tool.
- So the materials start removing from the workpiece as positive from the workpiece are dissolved and carried away by electrolyte and materials are also removed due to the striking power of the negative ions coming from the tool.
- This material removal process from the workpiece is continued until a cavity is created in the workpiece, a replica of the tool used in this process.
- So, In this way material is removed in the electrochemical machining process.
Fig,-Electrochemical machining process diagram-

Advantages of electrochemical machining-
Here are some electrochemical machining advantages, given below-
- It can cut any material irrespective of its hardness, strength, or toughness.
- Complex cavities can be produced using ECM machining.
- Tool wear is zero as there is no direct contact between the tool and the workpiece.
- A very good surface finish is produced in this process as the material removal takes place due to atomic level dissociation.
- Very little heat is generated during this process.
- No residual stresses as there is no direct contact between the tool and the workpiece.
- There is no thermal damage to the workpiece.
Disadvantages of electrochemical machining-
Here are some of the electrochemical machining disadvantages, given down-
- Only electricity-conductive material can be machined.
- It can not be used for machining soft materials and non-metals.
- The original problem of poor dimensional accuracy
- Environmentally polluting large waste
- The acidic electrolyte poses the risk of corrosion to tools, workpieces, and equipment.
- High energy consumption
Electrochemical machining Applications-
Some of the electrochemical machining uses, again given down-
- Sinking operations
- Drilling
- Trepanning
- Profile and contouring
- Micro-machining
Process Parameters of electrochemical machining-
Here are the electrochemical machining process parameters, given below-
| Power- | |
| Voltage | 2 to 35V |
| Current | 50 to 40,000 A |
| Current Density | 0.1 A/mm2 to 5 A/mm2 |
| Electrolyte- | |
| Material | NaCl and NaNo3 |
| Temperature | 20°C to 50°C |
| Flow Rate | 20 Ipm per 100 A current |
| Dilution | 100 g/l to 500 g/l |
| Working Gap | 0.1mm to 2mm |
| Overcut | 0.2mm to 3mm |
| Feed Rate | 0.5mm/min to 15mm/min |
History of Electrochemical machining-
Russian scientist E. Shpitalsky‘s 1911 invention of electrolytic polishing served as the basis for the development of electrochemical machining (ECM).
The first use of ECM was by W. Gusseff in 1929; since then, its industrial applications have expanded to include electro-chemical drilling, electrochemical deburring, electrochemical grinding, and electrochemical polishing.
Additionally, in the 1960s and 1970s, as a machining technique.
A relatively old machining method, the foundations of precise electrochemical machining were established in the 19th century.
Since then, the process has been improved, making it an incredibly accurate method with a wide range of potential applications.
Wrapping Up-
That’s it for the Electrochemical machining process, Where you learn about various topics like definition, working principle, various equipment, the electrolyte used, working, process parameters, advantages, disadvantages, and applications.
I hope, this article added some value to you and fulfilled your goal of landing here. If so, consider sharing with your nearby, colleagues, friends, and family or to the needed ones.
Bookmark to Engineers Rail to ease the excess further. Thank you for being with me. I hope to see you again.
Read Also-
Electrical Discharge Machining
Water Jet Machining: Definition, Principle, Working, Parameters, Advantages, Applications
Before you go, Here are your freebies all yours-
| Electrochemical machining ppt |  |
| Electrochemical machining pdf |

Abhishek Tiwary is a blogger by passion and a Quality Engineer by profession. He completed his B.Tech degree in the year 2017. Now working in a reputed firm. He loves to share his knowledge with others.



